In this article, learn more about the derivation of the formulas and equations describing the isentropic (adiabatic) process.
Basic equations
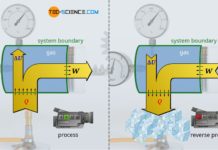
For the derivation of the equations describing the isentropic process, the first law of thermodynamics is used with the restriction that in an isentropic process, by definition, no heat is transferred (Q=0):
\begin{align}
&\boxed{W_\text{V} + Q = \Delta U} ~~~~~&&\text{First Law of Thermodynamics}\\[5px]
\label{wv}
&W_\text{V} = \Delta U ~~~~&&\text{only applies to an isentropic process where }Q=0\\[5px]
\end{align}
The pressure-volume work Wv is determined by integrating the pressure function with respect to the volume (Wv=-∫p⋅dV), whereas the change in internal energy ΔU is determined by the temperature difference (ΔU=cv⋅m⋅ΔT). Thus, the following relationship applies between the change in volume and the change in temperature in an isentropic process:
\begin{align}
&W_\text{v} = \Delta U \\[5px]
\label{eq:5561}
&\boxed{- \int p~\text{d}V =c_\text{v}~m~\Delta T} ~~~~~\text{integral form }\\[5px]
\end{align}
If only infinitesimal changes in volume and temperature are considered at this point (within which the pressure can be considered constant), the pressure-volume work and the change in internal energy can be expressed by the respective differentials (Δ→d):
\begin{align}
\label{eq:9887}
&\text{d}W_\text{V} = \text{d}U \\[5px]
\label{pv}
&\boxed{-p~\text{d}V = c_\text{v}~m~\text{d}T} ~~~~~\text{differential form}\\[5px]
\end{align}
Relationship between volume and temperature
Equation (\ref{pv}) provides the relationship between an infinitesimal volume change dV and the resulting temperature change dT for the isentropic process. However, the pressure p is not independent of the volume V or the temperature T, as can be seen from the ideal gas law:
\begin{align}
\label{0}
&p~V=R_\text{s}~m~T ~~~~~\text{ideal gas law} \\[5px]
\label{p}
&\boxed{p=R_\text{s}~m~{T \over V}} \\[5px]
\end{align}
If the pressure function according to equation (\ref{p}) is used in equation (\ref{pv}), then the variables belonging to their differential can be separated (called separation of variables):
\begin{align}
\require{cancel}
– \overbrace{R_\text{s}~\bcancel{m}~{T \over V}}^{=p}~\text{d}V &= c_\text{v}~\bcancel{m}~\text{d}T \\[5px]
\label{tv}
– R_\text{s}~{1 \over V}~\text{d}V &= c_\text{v}~{1 \over T}~\text{d}T \\[5px]
\end{align}
Now that the variables are separated, this equation can be integrated within the respective limits V1 to V2 and T1 to T2:
\begin{align}
\label{eq:9552}
– R_\text{s}~\int\limits_{V_1}^{V_2} {1 \over V}~\text{d}V &= c_\text{v}~\int\limits_{V_1}^{V_2} {1 \over T}~\text{d}T \\[5px]
R_\text{s}~\left[ \ln{(V)} \right]^{V_2}_{V_1} &= c_\text{v}~\left[ \ln{(T)} \right]^{T_2}_{T_1} \\[5px]
– R_\text{s}~\ln \left(V_2 – V_1 \right) &= c_\text{v}~\ln \left(T_2 – T_1 \right) \\[5px]
R_\text{s}~\ln \left(V_1 – V_2 \right) &= c_\text{v}~\ln \left(T_2 – T_1 \right) ~~~\text{where}~~~ \underline{\ln(a)-\ln(b)=\ln \left(a \over b \right)} ~~~\text{:} \\[5px]
R_\text{s}~\ln \left(V_1 \over V_2 \right) &= c_\text{v}~\ln \left(T_2 \over T_1 \right) ~~~\text{where}~~~ \underline{R_\text{s}=c_\text{p}-c_\text{v}} ~~~\text{:} \\[5px]
(c_\text{p}-c_\text{v})~\ln \left(V_1 \over V_2 \right) &= c_\text{v}~\ln \left(T_2 \over T_1 \right) \\[5px]
\label{fre}
\left(\frac{c_\text{p}}{c_\text{v}}-1\right)~\ln \left(V_1 \over V_2 \right) &= \ln \left(T_2 \over T_1 \right) \\[5px]
\end{align}
At this point, the ratio of the specific heat capacities cp/cv form a new constant, the so-called adiabatic index κ. This constant serves at this point only the simpler notation!
\begin{align}
&\boxed{\kappa:=\frac{c_\text{p}}{c_\text{v}}} ~~~\text{heat capacity ratio (adiabatic index)}\\[5px]
\end{align}
With this definition of the adiabatic index, equation (\ref{fre}) can be written as follows:
\begin{align}
\label{k}
&(\kappa – 1)~\ln \left(V_1 \over V_2 \right) = \ln \left(T_2 \over T_1 \right) \\[5px]
\end{align}
Equation (\ref{k}) already gives a clear relationship between any initial state 1 (V1, T1) and any final state 2 (V2, T2) for the isentropic process. With the help of different logarithm rules, however, this equation can be simplified:
\begin{align}
\label{eq:3049}
e^{(\kappa-1)\ln \left(V_1 \over V_2 \right)} &= e^{\ln \left(T_2 \over T_1 \right)} \\[5px]
\left[e^{\ln \left(V_1 \over V_2 \right)}\right]^{\kappa-1} &= e^{\ln \left(T_2 \over T_1 \right)} \\[5px]
\label{w}
\left(V_1 \over V_2 \right)^{\kappa-1} &= {T_2 \over T_1} \\[5px]
\end{align}
\begin{equation}
\boxed{T_1 \cdot V_1^{\kappa-1}= T_2 \cdot V_2^{\kappa-1}} ~~~\text{bzw.}~~~ \boxed{T \cdot V^{\kappa-1}=\text{constant}}
\label{eq:5377}
\end{equation}
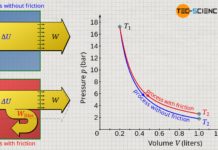
Finally, for an isentropic process the product of temperature and the volume exponentiated by κ-1 is constant!
Relationship between temperature and pressure
If the general relationship of two states obtained from the ideal gas law is used at this point, a clear relationship between the temperatures and the pressures can also be found:
\begin{align}
\label{a}
&\boxed{{p_1~V_1 \over T_1}={p_2~V_2 \over T_2} }\\[5px]
&{V_1 \over V_2} = {p_2 \over p_1}~{T_1 \over T_2} \\[5px]
\end{align}
If this ratio of volumes is used in equation (\ref{w}), the following relationship is obtained between the temperatures and the pressures:
\begin{align}
&{\underbrace{\left({p_2 \over p_1}~{T_1 \over T_2}\right)}_{\frac{V_1}{V_2}}}^{\kappa-1} = {T_2 \over T_1} \\[5px]
&\left(p_2 \over p_1\right)^{\kappa-1}~\left(T_1 \over T_2 \right)^{\kappa-1} = {T_2 \over T_1} \\[5px]
&\left(p_2 \over p_1\right)^{\kappa-1} = {T_2 \over T_1}~\left(T_2 \over T_1 \right)^{\kappa-1} \\[5px]
&\left(p_2 \over p_1\right)^{\kappa-1} = \left(T_2 \over T_1 \right)^{\kappa} \\[5px]
&\left(p_1 \over p_2\right)^{1-\kappa} = \left(T_2 \over T_1 \right)^{\kappa} \\[5px]
& \boxed{T_1^\kappa \cdot p_1^{1-\kappa} = T_2^\kappa \cdot p_2^{1-\kappa}} ~~~\text{bzw.}~~~ \boxed{T^\kappa \cdot p^{1-\kappa}=\text{constant}} \\[5px]
\end{align}
Relationship between volume and pressure
In the analogous way, a relation between volumes and pressures can be derived if equation (\ref{a}) is first solved for the ratio of temperatures:
\begin{align}
&\boxed{{p_1~V_1 \over T_1}={p_2~V_2 \over T_2} }\\[5px]
&\frac{T_2}{T_1} = {p_2 \over p_1}~{V_2 \over V_1} \\[5px]
\end{align}
If this ratio of temperatures is used in equation (\ref{w}), the following relationship is obtained between the temperatures and the volumes:
\begin{align}
&\left(V_1 \over V_2 \right)^{\kappa-1} = \underbrace{{p_2 \over p_1}~{V_2 \over V_1}}_{\frac{T_2}{T_1}} \\[5px]
&\left(V_1 \over V_2 \right)~\left(V_1 \over V_2 \right)^{\kappa-1} = {p_2 \over p_1} \\[5px]
&\left(V_1 \over V_2 \right)^{\kappa} = {p_2 \over p_1} \\[5px]
&\boxed{p_1 \cdot V_1^\kappa = p_2 \cdot V_2^\kappa} ~~~\text{bzw.}~~~ \boxed{p \cdot V^\kappa=\text{constant}} \\[5px]
\end{align}
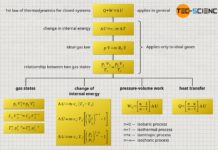
Summary of the equations
Finally, for an isentropic process, all the relationships between the various state variables were derived and are summarized again below:
\begin{alignat}{3}
&p \cdot V^\kappa = \text{constant}& &\Rightarrow&~ &\boxed{ p_1~V_1^\kappa = p_2~V_2^\kappa} \\[5px]
\label{3}
&T \cdot V^{\kappa-1} = \text{constant}& &\Rightarrow&~ &\boxed{ T_1~V_1^{\kappa-1} = T_2~V_2^{\kappa-1}} \\[5px]
\label{4}
&T^\kappa \cdot p^{1-\kappa} = \text{constant}& &\Rightarrow&~ &\boxed{ T_1^\kappa~p_1^{1-\kappa} = T_2^\kappa~p_2^{1-\kappa}} \\[5px]
\label{1}
&&&&&\text{ where } \boxed{\kappa = {c_\text{p} \over c_\text{v}}}>1 \\[5px]
\end{alignat}